
A case study for assay-ready patient-derived organoids (PDOs) and high-throughput 3D imaging to advance drug discovery
Introduction – the problem.
The average cost of bringing a new drug to the clinic is around $1 billion according to a study conducted by the London School of Economics in March 2020 (www.lse.ac.uk). This is partly due to the poorly representative models used for screening in the early stages of drug discovery, leading to a high failure rate of compounds later in the drug development pipeline. This has driven the search for more patient-centric models for early, accurate selection of lead candidates for further clinical development.
Patient-Derived Organoids (PDOs or just “organoids”) grown in three dimensions represent a solution to this problem. They are miniature copies of the normal or diseased human biopsy tissue from which they are derived and fully represent 3D tissues in the human body. Evidence shows that when patients and their derived organoids are treated with the same drugs, they show a mirrored response (Vlachogiannis et al., 2018). This demonstrates the proof of principle that organoids can be used for screening “libraries” of potential therapeutics. Compounds that have the desired effect on patient organoids are likely to be effective in treating the patients themselves.
What are Patient-Derived Organoids (PDOs)?
- Derived from normal or diseased patient biopsy tissue
- Grown in 3D in a protein gel with liquid feed
- Formed from multiple cell-types (as are organs in the body)
- Miniature organ replicates (e.g., "mini guts")
- Fully representative of human biology
- Patent "avatars" for use in drug discovery
- Grown in a bio-processor to create millions of standardized copies per batch

Bright-field image of 3D, colorectal cancer, patient-derived organoids (PDOs)
Ideally, once drugs have been properly tested and licensed for use in the clinic, individual drug treatments would be designed for each patient, based on the response of their own organoids. Unfortunately, to derive and expand organoids currently takes several weeks in the lab. Clinicians would not wish to wait that long before starting therapy. Personalised therapy may be possible in the future as the technology for deriving, culturing, and expanding organoids is further refined and developed.
3D organoids are a relatively new technology and assays to make full use of them are still being invented and developed. Researchers are more used to working with 2D cell monolayers that are much less complex, but do not accurately represent human biology.
The challenge - accurate selection of compounds from early screens, for further development and clinical testing.
The viability assay is one common method to quantify compound effectiveness (“efficacy”). One frequently used technique is a bioluminescent-based assay which detects the presence of metabolically active cells by measuring ATP levels. High-content imaging can also be used to determine cell viability by counting the number of live or dead cells (labelled with dyes such as Calcein AM, a common live cell marker) in a drug-treated sample in comparison to an untreated control (and possibly organoids derived from non-cancer-tissue). The desired effect of drugs used in therapy is to kill as many cancer cells as possible, whilst leaving the healthy tissue unaffected. The number of cells that are still alive in the untreated sample will be considerably more than in the drug-treated cells, especially where high concentrations of the therapy are used. It is therefore important to quantify the difference between the two conditions and to determine if anti-cancer compounds such as Trametinib have efficacy. The same principle can be used to identify new therapeutics that are working effectively to kill cancer cells as part of a screen from a library of compounds.
Not all drugs act in the same way. Some drugs act specifically on a targeted subset of cells or alter signals within the cells to prevent regrowth and spread of the disease. There is no bulk cell-killing effect, so the differences between healthy and cancer cells are not easily distinguished or quantified by counting the number of live cells remaining after treatment. Alternative methodologies are needed.
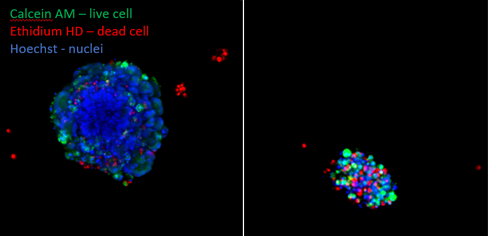
Colorectal cancer organoids were left untreated (A) or exposed up to Trametinib (5mM) (B). They were stained after 5 days to show up the live cells (green, Calcein AM) and dead cells (red, ethidium homodimer). It can clearly be seen that there are very few live cells remaining after the Trametinib treatment.
The solution – to detect the subtle effects of drugs acting on specific targets through high-throughput 3D imaging
Scientists have observed that the treatment of organoids with drugs can result in a marked change of appearance (morphology) which is related to the effect of the drug on the cells. In addition to the viability, other fluorescent markers can be added to the sample to obtain cell and organelle specific information – such as cytoskeleton structure, mitochondria, and overall organoid morphology. Capturing images of organoids with high-content imaging and using data analysis tools to quantify changes, can therefore potentially be used to indicate when an unknown compound is having an effect. This is especially relevant where a live/dead cell assay cannot detect any difference between treated and untreated cells. (Badder et al., 2020).
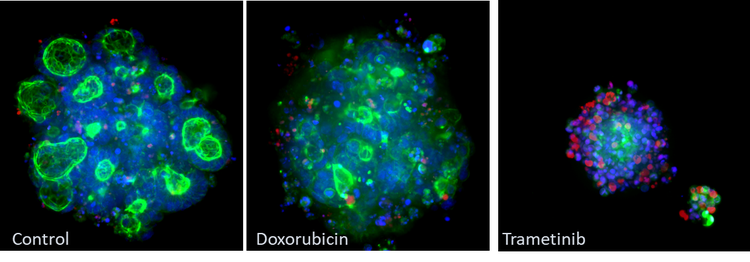
Example of morphological changes captured using Molecular Devices ImageXpress® Micro Confocal system. CRC organoids were fixed stained with phalloidin (green), Hoechst (blue) and ethidium homodimer (red). Note the changes in the phalloidin stain (green) between the control and treatment groups
To further demonstrate the principle that 3D organoid imaging can be used in drug discovery, Cellesce used Molecular Devices’ state-of-the-art automation, imaging technology, and advanced analysis of the 3D imaging data sets to compare the physical characteristics of untreated and drug-treated colorectal cancer organoids. Artificial intelligence-aided image analysis tools were used to analyze the CRC images and various phenotypic descriptors were used to quantify the effects of the treatment. An example of one of the measurements that can be made, i.e. total area, is shown below.
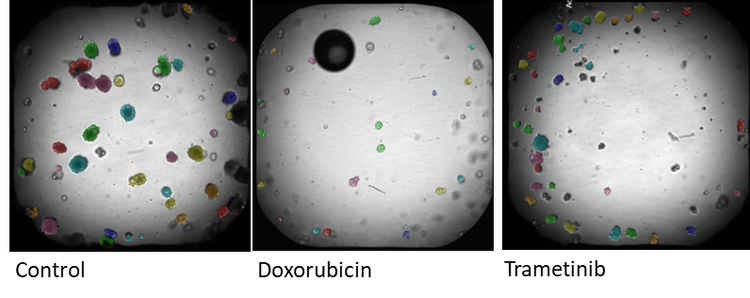
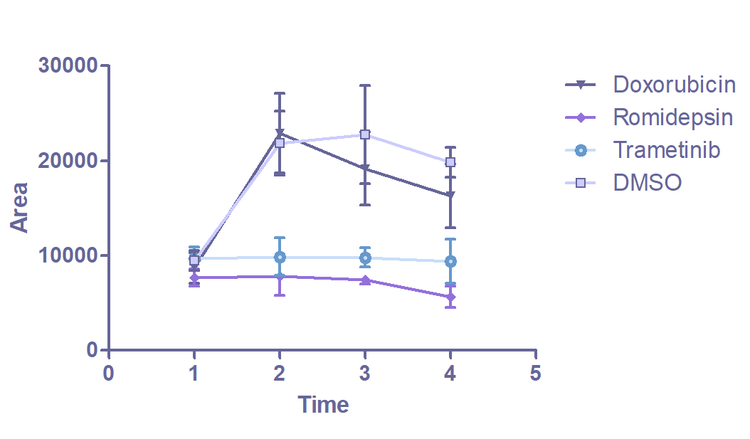
Images of organoids captured on the ImageXpress Confocal. Brightfield imaging is used to monitor changes in organoids morphology over time. Analysis is carried out in IN Carta image analysis software. Each identified organoid is overlaid with a colored mask. The graph (E) shows the change in average organoid area over 5 days.
The greatest reduction in total area is seen in organoids treated with the drugs romidepsin and trametinib. Compared to the control, treatment with romidepsin and trametinib arrested the growth of the organoids. This result is consistent with data from the viability assay, where romidepsin- and trametinib-treated organoids show a significant increase in dead cells (as a proportion of the total number of cells) compared to the untreated control. Initial analysis of the imaging data confirmed the relationship between organoid morphology and drug response. This demonstrated the proof of principle that this technique can be used in drug screens.
The use of organoids and 3D imaging has the potential to revolutionize early screening in drug discovery. This requires standardized and repeatable batches of large numbers of organoids. Since organoids are cultured manually, this requirement cannot be fulfilled easily. Cellesce has addressed this need through the development of a patented bioprocess. This novel, unique technology, uses bio-processors for the controlled production of sufficient quantities of organoids for high-throughput screening. Molecular Devices is addressing the additional requirements for automating sample handling using robotics and assay optimization using imaging and data analysis.
The use of assay-ready organoids, together with automation of high-throughput screening and quantification assays will facilitate accurate selection of candidate therapeutics from large compound libraries. This will go a long way towards the process of identifying effective treatments and accelerating the drug discovery pipeline. The accurate selection of potential therapeutic compounds in the early stages of drug discovery will reduce resource waste and the cost of development. This will lead to an increase in the number of drugs on the market. Clinicians will thus be able to choose directed treatments that are the most suitable for each patient, with minimal adverse side effects. This will improve quality of life and increase survival rates.
Improving drug development: Molecular Devices and Cellesce aim to advance the use of organoids
The recent acquisition of Cellesce affirms Molecular Devices’ commitment to investing in 3D biology technologies that transform the drug discovery process and drive development of novel therapeutics.
“Molecular Devices has the capability, reputation, reach, and resources to ensure that the Cellesce technology can be further developed, and used to its full potential,” said Vicky Marsh-Durban, CEO of Cellesce. “We’re excited to bring our domain expertise and intellectual property to Molecular Devices, together maximizing impact for customers in revolutionizing drug discovery and unlocking the full potential of human-relevant 3D biology research.”
Dive deeper into the conversation with Tanya Samazan from Instrument Business Outlook and Molecular Devices President Susan Murphy and Cellesce CEO Vicky Marsh-Durban.

Download our Organoid eBook
If you're ready to add another dimension to your research, The essential guide to organoids in drug discovery eBook delves into the history, current role, and future impact of organoids in drug discovery and how you can successfully integrate organoids into your research.