
Application Note
Comparison of intracellular calcium measurements with FLIPR Calcium Assay Kit
- Reduce cell detachment and diminished calcium signal response associated with dye loading and washing procedures with mix-and-read assay format
- Address real-time kinetic cell-based assays in multiple formats
- Configure assay parameters easily with user-friendly ScreenWorks Software
Introduction
G-protein coupled receptors (GPCRs) play an important role in cell signaling (Figure 1). When the receptor is activated by a ligand, receptor conformation is changed, triggering G-protein activation inside the cell. An active G-protein has the potential to induce various cascades of intracellular messengers including calcium. The FLIPR® System performs high-throughput, functional cell-based assays and is the system of choice in drug discovery for evaluating changes in intracellular calcium detected through use of fluorescent calcium-sensitive reporter dyes. This application note provides a basic protocol for performing a calcium mobilization assay using the FLIPR Calcium Assay Kit; a homogeneous, fast and reliable fluorescence assay on the FLIPR Tetra® System. The FLIPR Tetra System is used to determine the dose-response of carbachol in CHO M1WT3 cells transfected with Rat Muscarinic Receptor (M1). Data from 96-, 384- and 1536-well microplate formats are presented.
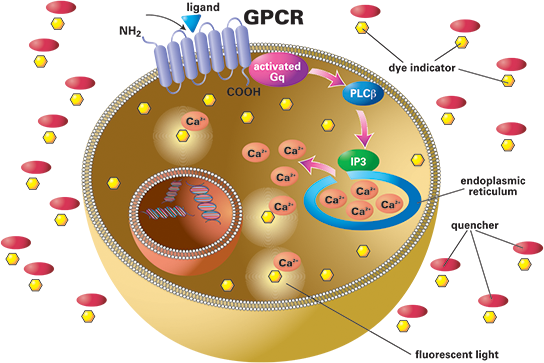
Figure 1. Calcium mobilization assay principle. Increase in cytosolic calcium can be detected by fluorescence measurement using calcium-sensitive dye indicators.
Materials
- CHO M1WT3 Cells transfected with rat Muscarinic (M1) receptor (American Type Culture Collection Cat. #CRL-1985)
- Culture media: Ham’s F12 Media (Irvine Scientific Cat. #9058) 10%, Fetal Bovine Serum (Hyclone Cat. #SH30071.03) 1%, Pen/Strep/Glutamine (Invitrogen, Cat. #10378-016), 50 µg/mL Geneticin (Invitrogen Cat. #10131-035)
- PBS without calcium and magnesium (Invitrogen Cat. #14190-144)
- VERSENE ® 1:5000 (Invitrogen Cat. #15040-066)
- Trypsin-EDTA 1X Solution (Irvine Scientific, Cat. #9341)
- 10X Hank’s Balanced Salt Solution (HBSS, Invitrogen Cat. #1406-5056)
- Sterile Water (Irvine Scientific, Cat. #9309)
- HEPES (Invitrogen Cat. #15630-080)
- Probenecid (Sigma Cat. #P 8761)
- FLIPR Calcium Assay Kit (Molecular Devices Cat. #R8033)
- AquaMax ® DW4 Dispenser and Washer (Molecular Devices)
- FLIPR Tetra Fluorometric Imaging Plate Reader (Molecular Devices)
Methods
Cell preparation
Step 1: Prepare cultures for assay. Split CHO M1WT3 cells 1:30, 2 times per week in culture medium and incubate at 37°C with 5% CO2
Step 2: Prepare cell plates. Begin by rinsing cell monolayer in flasks with PBS without calcium and magnesium.
Step 3: Incubate flasks 5 minutes at 37°C in VERSENE 1:5000 (or Trypsin-EDTA) and remove cells by gently rinsing with serum containing culture media and tapping the side of the flask.
Step 4: Centrifuge cells at 400 x g for 5 minutes. Re-suspend in culture media and count. Seed cells in black-wall clearbottom microplates and incubate overnight at 37°C and 5% CO2. Recommended cell concentrations and well volumes are listed in Table 1. A Molecular Devices AquaMax DW4 was used to dispense cells into 1536-well microplates.
Table 1. Plate format and cell number for FLIPR Calcium Assay Kit using the FLIPR Tetra System.
To observe changes in intracellular calcium levels, cells must be loaded with a calcium sensitive fluorescent dye. The simple and reliable “mix-and-read” assay format of Molecular Devices FLIPR Calcium Assay Kit substantially reduces or entirely eliminates cell detachment and diminished calcium signal response often associated with dye loading and washing procedures. Fluorescent signal from unincorporated extracellular dye is attenuated by patented quench-based technology (QBT™) resulting in a high signal-to-background ratio.
FLIPR Calcium Assay Kit protocol
Step 1: Start Loading Buffer preparation by dilution of 100 mL 10X HBSS in 880 mL sterile water for injection.
Step 2: Add 20 mL of 1M HEPES. Final concentration is 20 mM.
Step 3: Prepare 250 mM probenecid (PBC) stock solution in 5 mL 1N NaOH and 5 mL loading buffer.
Step 4: Prepare Loading Buffer + 5 mM PBC for 96-well and 384-well assays by adding 2 mL of 250 mM PBC to 98 mL loading buffer. For 1536-well assays, prepare Loading Buffer + 7.5 mM PBC by adding 2.01 mL PBC to 67 mL loading buffer
Step 5: Adjust pH to 7.4.
Step 6: To prepare Dye Loading Buffer, dissolve contents of one bulk-kit vial completely in the loading buffer with PBC made in Steps 4 and 5 that corresponds to assay well size being performed. Dissolve dye in vial first by vortexing with 10 mL loading buffer with PBC.
Step 7: Remove cell plates from the incubator and add appropriate volume of Dye Loading Buffer to each well. See Table 2 for the recommended volumes.
Note: Dye Loading Buffer for 15 1536-well microplates is added to microplates with an AquaMax DW4 System or equivalent.
Table 2. Recommended Calcium Assay Kit dilution volumes.
Step 8: Incubate plates for one hour at 37°C in 5% CO2, then at room temperature for 10 minutes.
Note: Plates are not washed prior to assay
Step 9: After incubation, transfer the plates directly to the FLIPR Tetra System and begin the calcium mobilization assay. For more information about dye preparations, cells and experimental parameters, refer to the FLIPR Calcium Assay Kit Product Insert or contact Molecular Devices Applications Support.
Compound plate preparation
Intracellular calcium measurements need to be performed quickly to visualize rapidly changing cellular kinetics. Optimal pipettor volumes, heights and speeds are easily configured on the FLIPR Tetra System. To deliver ligand to the cells without disturbing the monolayer, fluid volumes are adjusted so that approximately 10–30% of the final volume is added. Ligand concentrations are adjusted accordingly. In this experiment, ligand is added at 20% volume in 96- and 384-well formats and at 14.3% in 1536-well format.
Note: It is recommended to dilute screening compounds that require DMSO for solubility to a final concentration in the assay of 1% or less.
Configuring the FLIPR Tetra System
Building on the FLIPR System’s capabilities, the FLIPR Tetra System pipette heads are now easily changed by the user. In contrast to the 96- and 384- heads, which utilize disposable pipette tips, 1536-well fluid transfer is accomplished with a pipettor that couples a stainless steel cannula tip block with Molecular Devices’ novel elastomeric technology. The ScreenWorks® Software has a user-friendly interface for easy configuration of assay parameters. Refer to the FLIPR Tetra System Users Guide for a detailed discussion of the recommended instrument setup, configuration and assay parameters. Assay volumes, heights and speeds are experimentally determined for each cell type to provide optimal mixing without dislodging cells from the bottom of the well. Refer to Table 3 for recommended settings used with CHO M1WT3 cells.
Table 3. FLIPR Tetra System parameters for FLIPR Calcium Assay Kit.
Results
At the beginning of the assay (i.e., prior to compound addition), a “signal test” is performed to check basal fluorescence of the dye-loaded cells. It is best to work with cellular basal fluorescence signal at 200 to 1500 RFU. Refer to the “Running a Signal Test” section in the FLIPR Tetra System Users Guide for more information. The FLIPR Tetra System and the FLIPR Calcium Assay Kit are used to determine the change in relative light units elicited by carbachol-induced calcium mobilization.
Figure 2 shows carbachol dose-response curves in three formats generated using the FLIPR Calcium Assay Kit on the FLIPR Tetra System. The EC50 for 96-well and 1536-well microplate assays were 17.9 nM and 18.0 nM, respectively. The EC50 for the 384-well microplate assay was 14 nM.
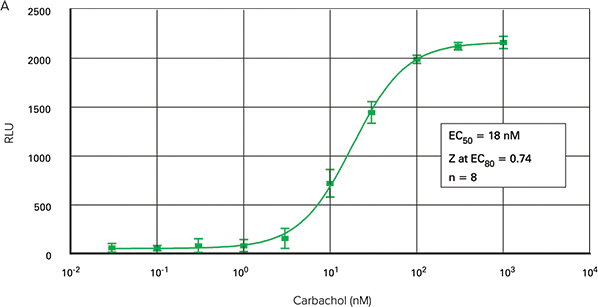
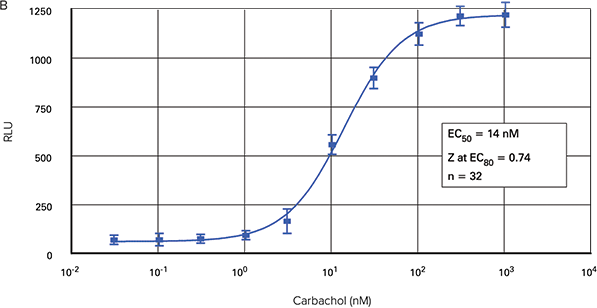

Figure 2. Carbachol dose-response curves. Carbachol-induced calcium mobilization in CHO M1WT3 cells plated in 96-well (A), 384-well (B) and 1536-well (C) microplates using the FLIPR Calcium Assay Kit on the FLIPR Tetra System.
The maximum amplitude of the fluorescent signal was greatest from the 96-well microplates. Those from the 384-well and 1536-well plates were very similar.
Z at EC80 values were > 0.70 for all three types of microplates. The signal trace for the fluorescent signal in response to carbachol in 384 wells is shown in Figure 3.
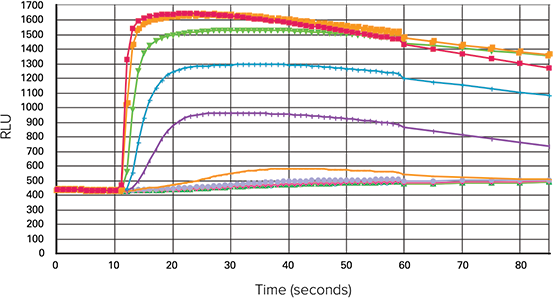
Figure 3. Average trace of carbachol in a 384-well microplate. Average well trace of a calcium mobilization assay stimulated by carbachol, generated on the FLIPR Tetra system.
Conclusion
The FLIPR Tetra System delivers a screening platform to address real-time kinetic cell-based assays in 96-, 384- and 1536-well format. Regardless of the FLIPR Tetra System microplate platform chosen, comparable results can be achieved, as evidenced by EC50 and Z values obtained in this study.