
Application Note
StainFree technology for cellular image analysis without fluorescent dyes
- Count cells and estimate confluence without fluorescent dyes
- Monitor cell growth without disrupting cells
- Set up imaging assays quickly and easily with the intuitive SoftMax Pro Software user interface
Introduction
Imaging cell-based assays typically requires the use of fluorescent probes that can be toxic to living cells or may only function in fixed cells. A label-free method for analyzing cell counts and cell confluence enables researchers to quantitatively monitor cell proliferation and health without time-consuming workflows that may disrupt cell viability. The SpectraMax® i3 Multi-Mode Microplate Reader with SpectraMax MiniMax™ 300 Imaging Cytometer uses patent-pending StainFree™ Cell Detection Technology. This allows users to perform cell proliferation, cytotoxicity, and other assays without nuclear stains like DAPI, which intercalates with DNA, or live cell dyes that are toxic to cells in the long term.
Here we demonstrate how StainFree technology enables cell counting that is comparable to nuclear and whole-cell counts obtained using fluorescent dyes. IC50 curves for compound-treated cells obtained through StainFree analysis are also presented as an alternative to using fluorescent reagents.
Counting cells with StainFree technology
CHO cells were plated at densities ranging from 8000 to 250 cells per well in a 384-well microplate and allowed to grow overnight. The following day, cells were imaged on the SpectraMax MiniMax 300 Imaging Cytometer with the transmitted light (TL) channel. To compare StainFree analysis to fluorescent cell counts, the same cells were then fixed and stained with green whole-cell or red nuclear dye and imaged using the corresponding fluorescence channels at 541 nm or 713 nm.
StainFree technology was used to count the cells in the TL channel. In the SoftMax® Pro Software, predefined analysis settings enable one-click results for a variety of cell types. Users can also use a supervised machine learning algorithm to easily ‘train’ the software to identify cells (Figure 1). Individual cells can be counted or confluence can be measured, offering a convenient way to monitor live cell growth at any time without having to stain the cells. Users also have the option to create customized settings as needed. Instrument temperature control can be used to maintain cells at 37°C during imaging for minimal disruption to cell viability.
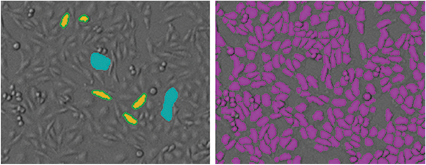
Figure 1. StainFree analysis in SoftMax Pro Software. Left: To create a new StainFree analysis setting, the mouse is used to ‘draw’ on the image, indicating individual cells (yellow) or non-cellular areas (blue). Right: A purple mask shows the objects identified in the image.
To compare fluorescent cell counts to the StainFree results, images acquired in the red and green fluorescent channels were also analyzed in the SoftMax Pro Software. Predefined settings for nuclear and whole cell counting are available in the software to accelerate analysis. Figure 2 shows that cell counts obtained with StainFree technology agree very closely with fluorescent nuclear and cytoplasmic cell counts.

Figure 2. StainFree technology compared to fluorescence cell counting. Cells counted using StainFree technology (blue), red nuclear stain (red), and green whole-cell stain (green). Counts obtained from all three methods agree closely, demonstrating that StainFree technology gives accurate cell counts while eliminating the need for fluorescent dyes (R2 > 0.99 for each plot).
Monitoring toxicity with StainFree technology
HeLa cells were plated at 5000 cells per well in 384-well microplates and allowed to grow overnight. Cells were then treated for 72 hours with several compounds that induce cell death through a variety of mechanisms and imaged on the MiniMax cytometer. Live cells were counted with StainFree analysis, and IC50 curves were plotted in the SoftMax Pro Software. Results confirm that cellular response to compounds can be monitored without disruptive staining and washing protocols (Figure 3).
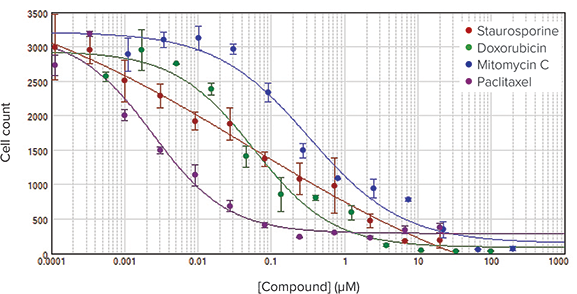
Figure 3. IC50 curves for cytotoxic compounds. Cell counts were performed using StainFree technology and plotted vs. compound concentration to obtain IC50 curves.
Conclusion
StainFree technology offers a new way to count cells and monitor cell growth without time-consuming and expensive staining procedures. Freedom from fixing or staining means that cells can be analyzed at any time without disrupting their growth, enabling better assay development. A user-friendly imaging interface in SoftMax Pro Software provides convenient predefined analysis settings for a variety of common cell types but also lets scientists customize their analysis as needed.