
Application Note
High-sensitivity ATP quantitation using SpectraMax Injector Cartridge with SmartInject Technology
- SmartInject Technology for complete reagent mixing with injection
- Sensitive detection of ATP down to 20 amol/well
- Dynamic range of 5 decades
Introduction
Adenosine 5´-triphosphate (ATP) is present in all living cells. Because the level is strictly controlled, the assay of ATP can be used as an indicator of viable cell number. ATP measurements are used to monitor raw materials and manufacturing plant hygiene for bacterial contamination in the food, drug, health care and personal healthcare industries as well as for waste water analysis.1 In the biotechnology and pharmaceutical industries, ATP measurements are used to evaluate cell proliferation and cytotoxicity. The bioluminescent assay of ATP has become very popular because of its high sensitivity and convenience. In this application note, we demonstrate highly sensitive detection of ATP down to 20 amol/well and a dynamic range of 5 decades using the SpectraMax i3x Multi-Mode Microplate Reader with SpectraMax Injector Cartridge and SmartInject™ Technology.
Assay principle
The basis for the assay is a reaction catalyzed by luciferase originally obtained from the common American firefly (Photinus pyralis).2 This enzyme catalyzes the ATPdependent oxidation of luciferin with the concomitant release of light (Figure 1). When ATP is the limiting component in the reaction, the amount of light emitted is proportional to the concentration of ATP. The intensity of the emitted light can be measured easily with the SpectraMax i3x reader. The SpectraMax Injector Cartridge enables precise reagent delivery and adjustment of delay and integration times that are required to obtain optimal sensitivity for this assay.
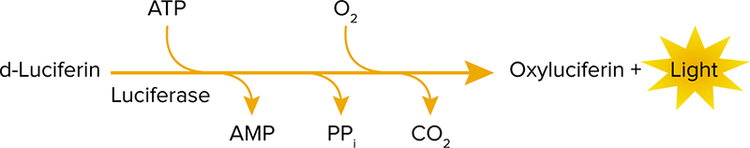
Figure 1. Reaction catalyzed by the firefly luciferase.
Materials and methods
- ENLITEN ATP Assay System (Promega cat. #FF2000)
- Solid white 96-well microplates (Greiner cat. #655075)
- SpectraMax i3x Multi-Mode Microplate Reader
Assay setup
The instructions supplied with the kit (Technical Manual Part #TMF004)3 were followed except for minor modifications to accommodate a 96-well format.
Note: The technical manual also includes a section discussing sample preparation including extraction of ATP from microorganisms or mammalian cells which is not covered in this application note.
- The enzyme and buffer solutions were thawed and kept on ice. The rL/L reagent was reconstituted by pouring the Reconstitution Buffer into the rL/L reagent vial, gently swirling several times and allowing it to stand at room temperature for one hour.
- For determining sensitivity and dynamic range, ATP standards were prepared by first diluting 100 µL ATP stock standard with 900 µL ATP-free water, followed by serial 1-to-10 dilutions in ATP-free water. The diluted standards (10 nM to 0.0001 nM) were kept on ice until use.
- Diluted ATP standards (or ATP-free water blanks) were pipetted into microplate wells in triplicate (10 µL/well). The final ATP content ranged from 0.01 to 1000 fmol/well.
Instrument setup
Instrument settings were selected using the Acquisition View in SoftMax Pro Software which provides an easily configurable graphical workflow (Figure 2). A preconfigured ATP assay protocol is also available in the software’s Protocol Library. The new SmartInject Technology was used to deliver reagent via the injector and simultaneously shaking the assay plate to ensure complete mixing of reagents.
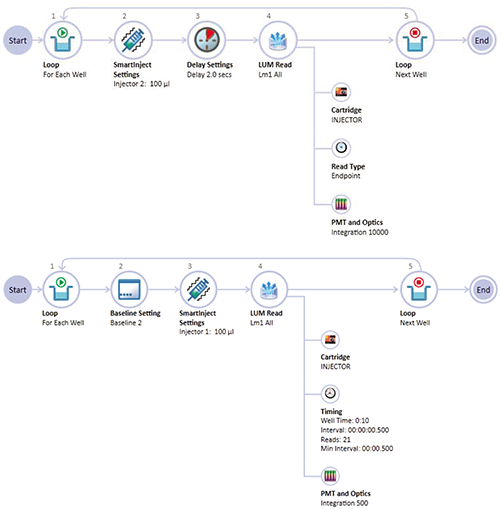
Figure 2. Acquisition Plan in SoftMax Pro Software. Top: Acquisition Plan (AP) for endpoint assay with 100-µL injection, 2-second delay, and 10-second (10,000 msec) integration time. This loop is repeated for each assay well in the plate. Bottom: AP for kinetic assay with 2 baseline reads, 100-µL injection, and kinetic read of 10 seconds with readings taken every 0.5 second using an integration time of 0.5 seconds (500 msec). SmartInject was used to deliver reagents while shaking the plate for complete reagent mixing.
- For a fast kinetic assay to determine the best settings for endpoint assays, SmartInject was selected to deliver 100 µL (injector 1) of rL/L reagent into selected wells containing ATP dilutions (10 µL/ well) and then to begin integrating at consecutive 0.5-sec intervals for a total of 10 seconds.
- For an endpoint read to determine sensitivity and dynamic range, 100 µL reagent was injected per well using the SmartInject Technology followed by a 2-second delay and a 10-second read.
- Injector 1 was primed with rL/L reagent by touching ‘Prime’ on the instrument touchscreen. This delivered 260 µL of reagent, sufficient for a full prime.
- The plate containing the ATP standards was placed into the plate drawer of the SpectraMax i3x reader and the injection/ read cycle was initiated by clicking the Read button.
Washing the injectors
Decontamination of the solvent delivery system is extremely critical for achieving satisfactory ATP assay results. Ethanol rinsing alone is not adequate. Not only must the system be sterilized but residual ATP from any source must be destroyed in order to maintain blank values as low as possible.
The Wash feature on the instrument touchscreen was configured to match required parameters for decontamination. Injector 1 was cleaned and sterilized by pumping at least 250 µL of 50% bleach (2.5%–3% sodium hypochlorite) through the injector and allowing it to stand for one hour, followed by washing with at least 2.5 mL of deionized water. Then at least 250 µL of 75% ethanol was pumped through the system and allowed to stand for one hour, followed by a deionized water wash of at least 2.5 mL. Finally, the injector was primed with air to clear all fluid from the line.
Results
ATP reaction profile
The reaction profile upon addition of the luciferin/luciferase reagent is shown in Figure 3. The light emission in the presence of 1000 fmol ATP/well (upper plot) was maximal within one second and remained at that level for at least 10 seconds. The blank (lower plot) showed essentially no response. Based on the data, a delay of two seconds is sufficient, and the integration time could be shortened to as little as one second, reducing the time required to read an entire plate.
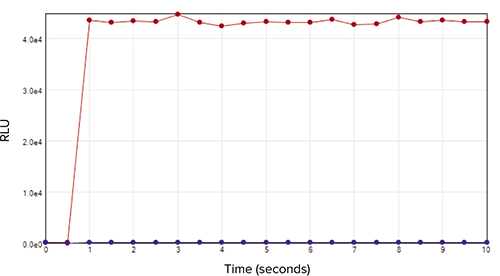
Figure 3. ATP reaction profile. Reaction profile for wells containing 1000 fmol/well ATP (red plot) or water only (blue plot) upon addition of luciferin/luciferase reagent is shown. Two baseline reads were taken followed by injection and additional reads taken at 0.5-second intervals. Data were analyzed and graphed in SoftMax Pro Software. Maximum signal was reached within 2 seconds of reagent addition and remained steady for the duration of the 10-second kinetic read. No appreciable response was seen for the water-only control well.
Dynamic range and sensitivity
A standard curve with ATP concentrations ranging from 0.01 to 1000 fmol/well is shown in Figure 4. The calculated limit of detection (amount producing a signal higher than 3 positive SD of the blank values above zero) was approximately 20 amol/well. The dynamic range was about 5 logs.
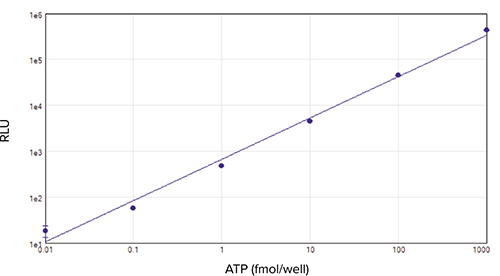
Figure 4. ATP standard curve. ATP standard curve obtained in a 96-well plate on the SpectraMax i3x reader with SpectraMax Injector Cartridge. Standards run in quadruplicate were analyzed and graphed using SoftMax Pro Software. Dynamic range is 5 decades, with r2 of 0.991.
Summary
ATP was detected down to 20 amol per well with a dynamic range of 5 decades, using the ENLITEN ATP assay system on the SpectraMax i3x reader with SpectraMax Injector Cartridge. This level of sensitivity allows users to reliably detect bacterial or other microbial contamination in foods, beverages, cosmetics, and other products as well as enzymes that degrade ATP.
Assay setup is simplified with a preconfigured protocol in SoftMax Pro Software that automatically plots a standard curve and analyzes sample data. The Acquisition View of SoftMax Pro Software lets users set up assay workflows easily using a graphical interface. SmartInject Technology provides simultaneous injection and mixing to ensure complete reagent mixing and rapid development of luminescent signal which are crucial for optimal assay sensitivity with minimal well-to-well variation.
References
- Jones, D. 1998. Bioluminescence assays using ATP. Luminescence Forum, 4: 1-9.
- DeLuca, M.A. and W.D. McElroy, (1978) in Meth. Enzymol., 53:3.
- Promega Corporation: www.promega.com