
Application Note
High-content screening of neuronal toxicity using iPSC-derived human neurons
- Set up and walk away with truly automated acquisition and analysis
- Instantly quantify multiple phenotypic features for each neuron
- Monitor real-time toxicity in live cells for minutes or days
Oksana Sirenko, PhD | Sr. Research Scientist | Molecular Devices
Introduction
The nervous system is sensitive to the toxic effects of many chemical compounds, environmental agents and certain naturally occurring substances. Neurotoxicity can cause temporary or permanent damage of the brain or peripheral nervous system during pathological processes such as spinal cord injury, stroke, or traumatic brain injury. It is also a major cause of neurodegenerative diseases such as Alzheimer’s and Parkinson’s disease.
High-content imaging using induced pluripotent stem cells (iPSCs) of human origin can be applied to examine neurotrophic, neuroprotective, or neurotoxic effects of pharmaceutical drug candidates or environmental contaminants. This note demonstrates neuronal toxicity screening via automated endpoint and live-cell assays using iPSCs and Molecular Devices systems and software.
iPSC-based toxicity screening
iPSCs are very useful for neuronal toxicity studies as they exhibit the functionality and behavior of mature neurons, and are also available in large quantities. This biologically relevant cell type paired with high-content imaging and analysis makes neurotoxicity assays valuable for screening lead compounds and potentially reduces pre-clinical development costs and the need for animal experimentation. The example assays presented were developed using fully differentiated human iPSC-derived iCell Neurons from Cellular Dynamics International.
Quantifying integrity of neuronal networks
High-content analysis provides a quantitative method to determine effects of positive and negative factors on neurite outgrowth (Figure 1). iPSC-derived neurons were allowed to form neuronal networks in 96- or 384-well plates for 3–5 days and then were challenged with toxic compounds for 48–72 hours. Neuronal networks were visualized with labeled antibodies against ß-tubulin on the ImageXpress® Micro System. Image analysis was performed using the Neurite Outgrowth Application Module in MetaXpress® High-Content Image Acquisition & Analysis Software, and data was analyzed using AcuityXpress™ High-Content Informatics Software.
The Neurite Outgrowth Application Module finds the cell body with or without a nuclear counterstain, determines a positive neuron cell via presence of a cell body and identifies the fluorescently-labeled neurites extending from those cells. Several parameters characterize the neuronal networks, including number of neurites, length of outgrowths, and number of branches per cell or per field. Statistics on number and phenotype of cells in each well can then be calculated.
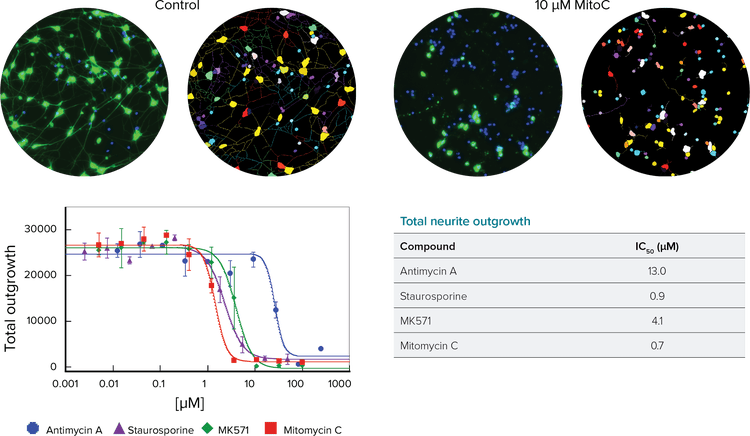
Figure 1. Neurite Outgrowth Application Module results. Top left: Control and high-dose wells with image analysis masks resulting from the Neurite Outgrowth Application Module. Nuclei (blue) and neurites labeled for ß-tubulin III (green). Top right: Dose-response curves to cytotoxic compounds for total outgrowth. Bottom: IC50 values calculated from total outgrowth curves.
Evaluating mitochondrial membrane potential
Mitochondrial depolarization has been shown to be an early sign of hypoxic damage or cell toxicity, and mitochondria membrane potential can be monitored using JC-10 dye (Figure 2). In healthy cells, JC-10 accumulates in mitochondria as orange J-aggregates. As the inner membrane potential is lost, the monomeric form of JC-10 is released into the cytoplasm where it fluoresces green.
Neurons were stained with JC-10 and exposed for 30 minutes to Antimycin A or Valinomycin, which are compounds that cause interruption of oxidative respiration and Ca2+ overload. The Granularity Module in MetaXpress Software can be customized to identify mitochondria based on intensity above background and size of the granular objects. Results were normalized to cell count in the field of view.
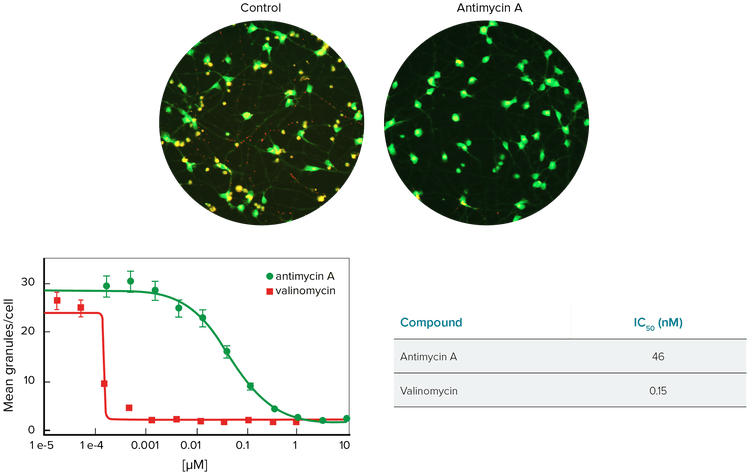
Figure 2. Neurons treated with Antimycin A. Top: Images of control neurons and neurons treated with Antimycin A and stained with JC-10. Images shown were acquired with 20X objective. Bottom: Dose response as measured by mean total mitochondria /granules per cell.
Monitoring live cell toxicity
Toxicity assays can be performed on live cells using a combination of the viability dye, Calcein AM, and Hoechst nuclear stain. A number of known neurotoxic and non-toxic compounds were tested using the ImageXpress Micro System with environmental control to maintain plate temperature, CO2, and humidity levels (Figure 3). The observed neurotoxic activity of these compounds correlated with in vivo or clinical data, confirming the potential for this assay to be used for predictive neurotoxicology.
Since Calcein AM stains the neuronal extensions in addition to the cell bodies of live neurons, neuronal networks were analyzed using the Neurite Outgrowth Application Module, and the live and dead neurons were identified both by Calcein AM staining and by nuclear size using the Cell Scoring Application Module.
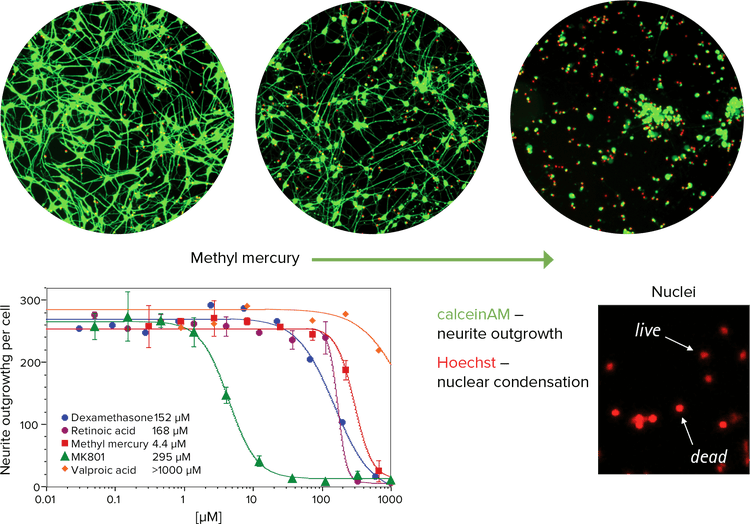
Figure 3. Effect of methyl mercury on neurons. Top: Images showing the dose-dependent toxic effect of methyl mercury on neurons. The presence of Calcein AM indicates metabolism of living cells (green). Bottom left: IC50 curves of cytotoxic compounds as determined by neurite outgrowth and evaluation of cytotoxicity by nuclear size. Bottom right: Hoechst stained nuclei are pseudocolored red.
A complete solution for neuronal toxicity screening
Molecular Devices offers a complete solution for evaluation of neuronal toxicity in a screening environment with the ImageXpress Micro System, MetaXpress software, and AcuityXpress software. Our live-cell option paired with sophisticated phenotypic image analysis enables identification of toxic compounds at an early stage of drug development.