
Application Note
Monitoring cytotoxicity on the SpectraMax i3 platform with MiniMax cytometer
- Get more biologically relevant data by combining microplate reader assays with imaging cytometry
- Visualize your cells to increase confidence in cell-based assay results
- Accessible to non-imaging experts with intuitive SoftMax Pro Software
- The only multi-mode platform that evolves to meet future application needs with user-installable application cartridges
Introduction
Apoptosis is an important process in embryonic development as well as in cancer and neurodegenerative diseases. Assays for apoptosis and cell viability can provide information on the mechanisms of cell death. Microplate reader assays often use whole-well luminescent or fluorescent readouts, but even more information can be gathered by combining these assays with imaging cytometry. The SpectraMax® i3 Multi-Mode Detection Platform allows users to measure apoptosis and cell viability on a well-by-well or cell-by-cell basis, as well as control assay quality by monitoring cells’ appearance.
Cell-based assay setup
HeLa cells were plated at 5000 cells per well in 384-well black-wall, clear-bottom microplates and allowed to grow overnight. The following day, a serial dilution of the apoptosis-inducing compound anisomycin was added to the cells. After 20 hours, apoptosis and cell viability were measured.
Apoptosis was monitored using the CellEvent Caspase 3/7 Green Detection Reagent (Life Technologies Cat. #C10423), which produces a green fluorescent signal in apoptotic cells where caspase-3 or caspase-7 has been activated. Apoptotic cells were detected using the SpectraMax® MiniMax™ Imaging Cytometer with cell count analysis. Cell viability was quantitated using the CellTiter-Glo Luminescent Cell Viability assay (Promega Cat. #G7570). This luciferase-based assay produces a luminescent signal proportional to the amount of ATP, and therefore metabolically active cells, in the assay wells. Luminescence was detected using the SpectraMax i3 platform. All data were collected and analyzed using SoftMax® Pro Software. Cells were also imaged using the bright field imaging mode of the MiniMax Imaging Cytometer for visual quality control.
Results
After 20 hours of treatment, anisomycin had induced apoptosis in a concentrationdependent manner, with apoptotic cells exhibiting green fluorescence (Figure 1).

Figure 1. Fluorescent imaging of caspase activity in HeLa cells. Top row: Fluorescent imaging of caspase activity in anisomycin-treated (left) and untreated control (right) cells. Apoptotic cells are green-fluorescent. Bottom row: Bright-field imaging of anisomycin-treated (left) and untreated (right) cells. The rounding of anisomycin-treated cells is consistent with apoptosis. All images were obtained with the SpectraMax MiniMax Imaging Cytometer.
Brightfield images showed rounding of anisomycin-treated cells, consistent with apoptosis. Caspase activity increased with increasing concentrations of anisomycin, while ATP levels declined (Figure 2).
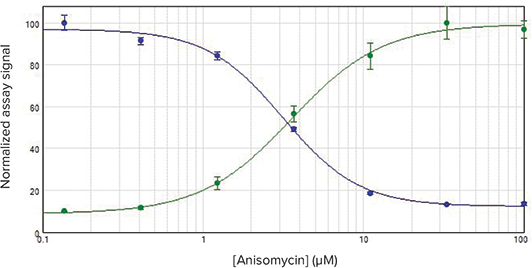
Figure 2. Normalized caspase activity and cell viability data. Caspase (green) and ATP assay (blue) results for anisomycin-treated HeLa cells. Caspase activity was detected using the SpectraMax MiniMax Imaging Cytometer, and ATP activity was measured using the luminescence detection mode of the SpectraMax i3 Multi-Mode Detection Platform.
Conclusion
A more complete assessment of cell health can be performed using a combination of fluorescence imaging and luminescencebased assays. Fluorescent imaging of caspase-3/7 activity, an essential event of apoptosis, was combined with a luminescent ATP assay to monitor cell viability. Brightfield images provided information on cell morphology that could be used for quality control.
The SpectraMax i3 platform with MiniMax Imaging Cytometer is an ideal platform combining the benefits of imaging with the versatility of multi-mode plate reader assays. SoftMax Pro Software offers a simple workflow from data collection to analysis, and user-upgradeable cartridge options make this system an intelligent investment.