
Application Note
Multiplexing Fluorescent and Chemiluminescent Live Cell Reporters to Dissect NFkB Signal Transduction Requirements

The chemiluminescent Ready-To-Glow Secreted Metridia Luciferase reporter system from Clontech Laboratories was employed to monitor NFκB response element activation by TNF-α, and was combined with Clontech’s fluorescence-based ZsGreen Proteasome Sensor. This combined assay was used to directly prove the requirement for active proteasomes in TNF-α-induced, NFκBdependent signaling using the multi-detection capacity of the SpectraMax® M5 Multi-Mode Microplate Reader from Molecular Devices. Both reporters were measured in a completely homogeneous manner, in the same well of a 96-well plate, without cell lysis.
NFkB Translocation is Proteasome-Dependent
The proteasome is a large protein complex that degrades proteins into short peptides in a ubiquitin-dependent manner. It plays a vital role in processes such as gene transcription, cell-cycle progression, DNA repair, cellular differentiation, viral infection, and oncogenesis.1 Proteasome activity has been implicated in the regulation of NFκB-based transcription: to activate NFκB upon TNF-α stimulus, IkB must be phosphorylated and then degraded by the proteasome. If proteasome degradation is blocked, NFκB activation is compromised.2–3 However, until now it has not been possible to monitor proteasome activity and NFkB signaling simultaneously, in the same sample.
The ZsGreen Proteasome Sensor
The Living Colors HEK 293 ZsGreen Proteasome Sensor Cell Line (Clontech Laboratories Cat. #631535) provides a simple and noninvasive way to monitor proteasome activity.4 This stably transfected cell line is based on the Proteasome Sensor Vector (Clontech Laboratories Cat. #632425), which encodes a fusion of the wild-type ZsGreen fluorescent protein5 with a proteasome targeting sequence.6 Normally, the resulting fusion protein, ZsProSensor-1, is rapidly degraded. However, when proteasome function is compromised, ZsProSensor-1 accumulates quickly, and can be easily monitored in a fluorescence microplate reader or by flow cytometry. (4, 7–8) The ZsGreen Proteasome Sensor has previously been used to successfully monitor proteasome activity in a quantitative fashion on the SpectraMax® M5 Microplate Reader.8
Secreted Metridia Luciferase
The Ready-To-Glow Secreted Luciferase is a chemiluminescent, enzyme-based reporter which utilizes Clontech’s sequence- and human codon-optimized secreted Metridia longa luciferase.9 Secreted Metridia luciferase is ideal for characterizing signal transduction pathways in a homogeneous, live cell format. The assay exhibits high fold induction, extended signal stability and intensity, and possesses the high sensitivity of an enzyme-based system.9–10 The assay’s high sensitivity and broad dynamic range generally eliminate the need for sample dilution.11
The SpectraMax M5 Multi-mode Microplate Reader
The SpectraMax M5 Microplate Reader is a benchtop, dual-monochromator, multidetection instrument with assay performance similar to dedicated single-mode readers. The reader has five detection modes: UV-visible absorbance, fluorescence intensity, time-resolved fluorescence, fluorescence polarization, and luminescence.12
Data collection, analysis, and management were performed with SoftMax® Pro Software from Molecular Devices, which provides these functions for all Molecular Devices benchtop microplate readers, allowing cross-plate analysis and custom calculations.13 Protocols for use with Ready-To-Glow are available directly from the pulldown menus in SoftMax Pro Software.
Observing Proteasome Status & NFκB Signal Transduction
To make the connection between proteasomal activity and NFκB-dependent signal transduction, the Proteasome Sensor Cell Line was transiently transfected with a reporter vector encoding Ready-To-Glow secreted Metridia luciferase driven by the NFκB response element. ZsGreen fluorescence was used to monitor the activity of the proteasomes, while Metridia luciferase chemiluminescence was used to measure the transcriptional activation of the NFκB response element via NFκB, upon addition of TNF-α. Since the Proteasome Sensor and Ready-To-Glow assays are both livecell assays and the SpectraMax M5 Microplate Reader functions in both fluorescence and chemiluminescence modes, the measurements were performed in a multiplex format on the same cells, in the same well.
Transfected cells were induced with TNF-α (25 ng/mL) in the presence or absence of the proteasome inhibitor ALLN (50 µM). The ZsGreen fluorescent signal was used to monitor the proteasomal activity of the cells; then the secreted Metridia luciferase substrate was added to the same well, and the chemiluminescent signal was measured.
In the absence of ALLN, only low levels of ZsGreen were observed, indicating that the proteasomes were active and degrading the ZsProSensor-1 protein. Upon TNF-α induction of cells that were not treated with the proteasome inhibitor ALLN, a high level of the Metridia luciferase reporter was detected in the media supernatant of transfected cells, indicating that the NFκB response element was active and driving the expression of secreted Metridia luciferase. (See Figure 1.)
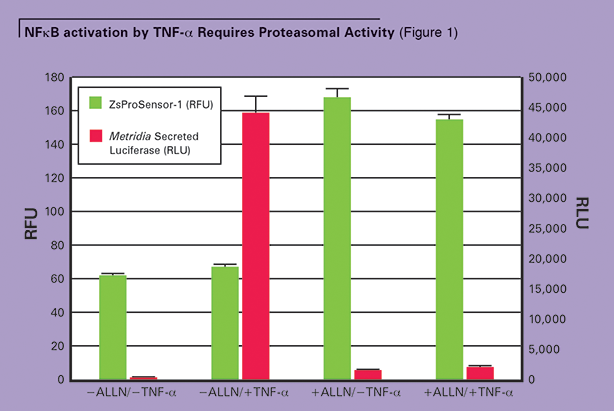
High levels of Metridia luciferase signal were only observed in the absence of ALLN and the presence of TNF-α. When the proteasome is inactivated by ALLN (monitored by increasing levels of ZsProSensor-1 fluorescence), the NFkB signaling pathway cannot respond effectively to TNF-α stimulation. ALLN = N-acetyl-leucyl-leucyl-norleucinal.
However, if cells were treated with TNF-α in the presence of ALLN, high levels of ZsGreen fluorescence were measured, indicating that the proteasomes were inactivated. Despite TNF-α induction, only low levels of the Metridia luciferase transcription reporter were detected, indicating that NFκB was not effectively activated (See Figure 1.) These combined results demonstrate the essential need for proteasomal activity in TNF-α-induced, NFkB-dependent signaling. (See Figure 2.)
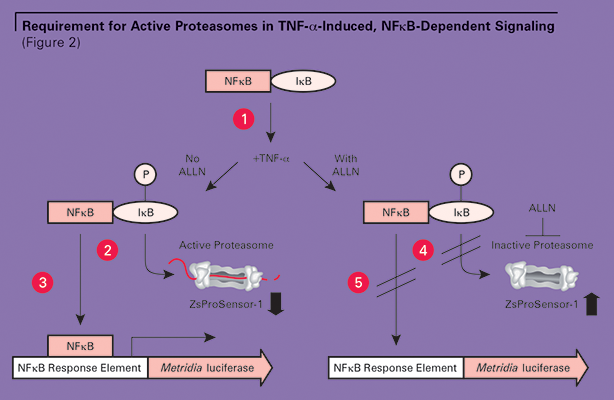
Inactive NFkB is sequestered in the cytoplasm by IkB. IkB must be phosphorylated upon TNF induction (1) and degraded by the proteasome (2) in order for NFkB to translocate to the nucleus and initiate signaling (3). Alternatively, when the proteasome is inhibited by the peptide ALLN (4), IkB is not degraded and NFkB cannot translocate (5). The status of the proteasome (active or inactive) can be monitored based on ZsProSensor-1 levels.
Conclusions
By combining the Proteasome Sensor with the Ready-To-Glow System, it was observed, in single cultures of live cells, that active proteasomes are required for NFκB to initiate transcription upon induction by TNF-α (Figure 2). Thus, the classical view that phosphorylation/dissociation of IkB is sufficient for NFκB signaling is incomplete.
The SpectraMax M5 Microplate Reader and accompanying SoftMax Pro software enabled the experiment to be unified as follows: immediately following the ZsGreen fluorescence measurement, the Metridia luciferase substrate was added to the same well and the resulting chemiluminescence measured. Both readings were captured from the same cells, on the same instrument, and with the same software. Thus, proteasomal activity and NFκB-dependent signaling induced by TNF-α were monitored simultaneously (Figure 1).
In the past, multiplex assay design was limited to (a) multiplexing different fluorescent proteins to create a live-cell assay, or (b) multiplexing different luciferases to create a lysis-based assay. However, since Metridia luciferase is a secreted reporter that does not rely on cell lysis, we were able to multiplex fluorescence- and chemiluminescence-based reporter systems for the first time, combining the advantages of both into one homogeneous, live-cell assay. Multiplexing fluorescent and chemiluminescent live-cell assays within the same well opens new avenues for intelligent assay development.
Acknowledgements
Clontech Laboratories would like to thank Cathy Olsen of Molecular Devices for her technical assistance.
References
- Adams, J. (2002) Trends Mol Med 8(4 Suppl):S49–S54.
- Haas, M. et al. (1998) J Leukocyte Biol 63(3):395–404.
- Sun, L. and Carpenter, G. (1998) Oncogene 16(16):2095–2102.
- Proteasome Sensor Vector. (April 2003) Clontechniques XVII(2):14.
- Matz, M. V. et al. (1999) Nature Biotech 17(10):969–973. Erratum in Nature Biotech 17(12):1227.
- Li, X. et al. (1998) J Biol Chem 273(52):34970–34975.
- Living Colors Cell Lines. (April 2004) Clontechniques XVII(2):14.
- Measurement of proteasome inhibition in live cells in Molecular Devices microplate fluorometers. (2006) SpectraMax Application Note. Molecular Devices.
- Ready-To-Glow Secreted Luciferase System. (July 2006) Clontechniques XXI(2):12–13.
- Ready-To-Glow Dual Secreted Reporter System. (October 2006) Clontechniques XXI(3):1.
- Detection Limit and Linear Range of Ready-To-Glow Secreted Metridia Luciferase Determined on the Molecular Devices SpectraMax L. (July 2007) Clontechniques XXII(3):20–21.
- SpectraMax M5/M5 e multi-detection reader: A five-mode microplate reader with threemode cuvette port. Molecular Devices Data Sheet (2006).
- SoftMax Pro Software: The industry standard in microplate data analysis. Molecular Devices Data Sheet (2006).