
Antigen / Immunogen Discovery and Optimization
Solutions for immunogen discovery and vaccine development
Vaccine development workflows vary widely depending upon the platform (e.g. inactivated virus vs. DNA vaccine) chosen, each having its own advantages. CEPI, the Coalition for Epidemic Preparedness Innovations, and many other organizations promote such diverse approaches during a pandemic in order to increase the likelihood of success against the infectious agent.
In this video, Justin Dranschack, manager, BioPharma platform, reviews our workflow solution for vaccine development using recombinant proteins as the immunogen, and references the systems to aid in your research.
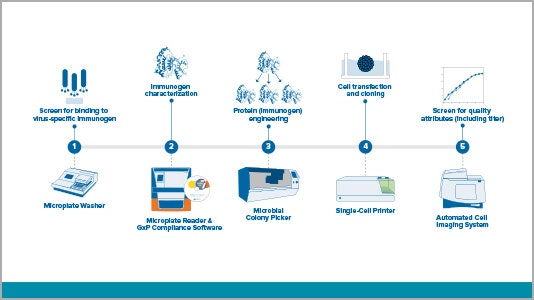
Antigen / immunogen discovery workflow

Step 1: Screen for binding to virus-specific antibody
Phage display is a powerful in vitro method that can be used to screen millions of antibodies against viral antigens, which are then considered as potential vaccine candidates.
Step 2: Immunogen characterization
Immunogenicity studies are conducted to determine whether the antigens identified in the previous step will initiate an immune response, since phage display only screens for antigen-antibody binding. If the antigen evokes an immune response (often measured by an ELISA), the antigen, now considered an immunogen, will move forward. Additional immunogen characterization is used to engineer desirable properties into the immunogen in the next stage.
Step 3: Protein (immunogen) engineering
Some properties of immunogens are known to cause undesirable effects when injected into animals or humans. Therefore, genetic engineering tools like CRISPR/Cas9 are used to manipulate the DNA encoding the immunogen so that they no longer possess those properties.
Step 4: Cell transfection and cloning
Once the DNA has been optimized, it is necessary to scale up the production of the immunogen for further testing. At this stage, it is critical to generate a high expressing, clonally derived cell line. This process often involves genetic engineering techniques described above along with single cell cloning into microwell plates, particularly if using mammalian cell lines.
Step 5: Screen for quality attributes including titer
Following single cell cloning, cell lines are monitored for growth and assessed for protein expression. The immunogen is then purified and prepared for injection into an animal in order to induce an immune response. Antibodies generated from the animal are then screened in a virus neutralization assay to determine vaccine efficacy.
COVID-19 related research solutions
Accelerate your COVID-19 research
Molecular Devices is committed to supporting scientists that are researching COVID-19 cellular response and vaccine development by offering technology and solutions that you can deploy rapidly.
Workflow solutions for vaccine development
View a variety of virus-related workflows – from antigent/immunogen and antibody discovery to stable cell line development.
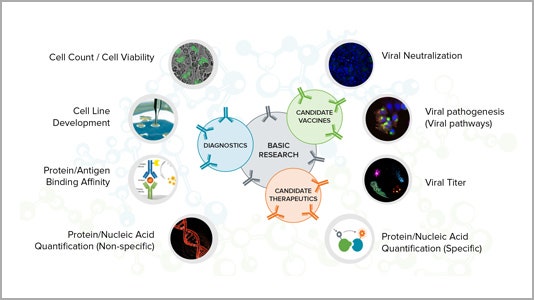
Applications for critical research
Learn more about how our technology can help your research of COVID-19 and other infectious diseases for vaccine development, therapeutics and diagnostics.
Resources related to COVID-19 cellular responses and vaccine development
Systems to accelerate your COVID-19 cellular response and vaccine development
We have validated and compliant laboratory solutions including microplate readers, microplate washers, biopharma and cellular imaging systems to meet your research needs.