
Application Note
Lung organoids as an assay model for in vitro assessment of toxicity effects by 3D high-content imaging and analysis
- Monitor and visualize the growth and differentiation of lung organoids using 3D imaging, reconstruction, and analysis
- Characterize and measure individual cells within each organoid
- Acquire images faster without sacrificing image quality with laser light source
Oksana Sirenko, PhD | Sr. Research Scientist | Molecular Devices
Angeline Lim, PhD | Applications Scientist | Molecular Devices
Mary Kassinos | Cell Culture Technician | Molecular Devices
Introduction
Organoid models have increasingly gained popularity in biologic research and screening to recapitulate complexity of real tissues. To model the in vivo human lungs, we have cultured primary human lung epithelial cells under conditions that promote the formation of 3D structures recapitulating the morphological and functional characteristics of the airway. In lung organoid cultures, epithelial stem and progenitor cells were cultured in ECM supplemented with a mixture of growth factors. The organoids then self-organized into complex structures retaining clusters of multi-lineage epithelial cells. These special characteristics make organoid culture a promising system for a wide range of applications in both basic and translational approaches such as drug screening and disease modeling.
We describe a high-content imaging method that monitors the growth and differentiation of lung organoids. 3D reconstruction allowed for further complex analysis of the organoid structure. Within the organoid, the cell morphology, viability status, and the expression of various cellular markers can also be measured. 3D lung organoids were embedded into Matrigel domes. Developing organoids comprised spherical objects with complex morphology including cavities and vesical structures. Increase in size and complexity was monitored during eight weeks of development. Organoids were monitored using transmitted light, then stained and imaged using 20–30 Z-planes through the Matrigel with 10X–40X magnifications. Using laser light source with new configuration of the ImageXpress® Confocal HT.ai High-Content Imaging System drastically increased the power of illumination that results in significantly increased brightness, assay sensitivity, and speed of imaging.
Methods
Cells: 3D lung organoids
3D lung organoids were derived from primary human lung epithelial cells (ScienCells, Co.). Cells were expanded for two weeks in 2D according to the protocol from ScienCells. Then the cells were seeded in 90% Matrigel (Corning) into Matrigel domes using reagents according to the protocol from Stem Cell Technologies (PneumaCult™ Airway Organoid Kit). Cells formed 3D organoids and were fed every second day for two weeks using PneumaCult Airway Organoid seeding media, then differentiated for another six weeks using PneumaCult Airway Organoid differentiation media. Organoids were grown in different plate formats (see Results section), and then treated with compounds or stained with various markers for morphology and viability assessment. The growth and development of organoids were monitored in transmitted light every week using 4X or 10X magnification. The schematic of the workflow is presented below:
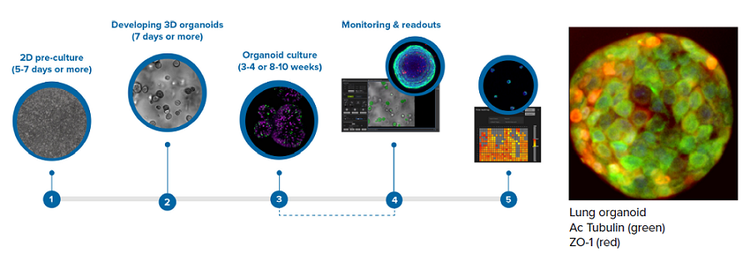
Results
Culturing and imaging 3D lung organoids
The culture was started from primary lung epithelial cells, and then organoids were grown in Matrigel domes using reagents and protocol from Stem Cell Technologies (see Methods section).
Briefly, cells were expanded in 2D, then mixed with GF-reduced Matrigel and seeded into Matrigel domes in 24-well plates or other plate formats.
- 2D expansion
- Matrigel dome 3D
- Differentiation
- Experiment
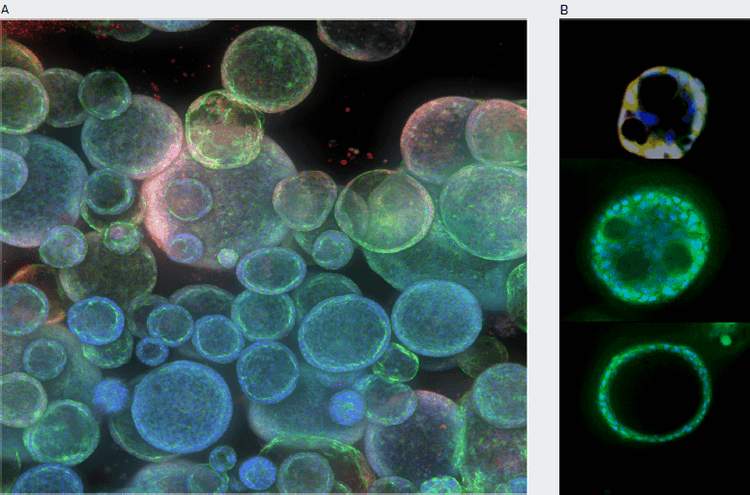
Figure 1. Monitoring complexity of organoid structure using confocal imaging. Live organoids were stained with Hoechst nuclear dye, MitoTracker, and Calcein AM. Confocal images were taken 5–10 μm apart, 10X or 20X. A. Maximum projection images. B. Single Z-confocal image shown allowing to observe and measure lumens, cavities, and protrusions, consistent with appearance of more mature phenotypes.
Monitoring organoid growth and 3D image analysis
Organoid growth and development were monitored using transmitted light imaging or using Hoechst stain. Automated imaging of live organoids in Matrigel was done using confocal option, 4X or 10X magnification. Image analysis of a single Z-plane or 3D projection was done to determine the approximate number and size of organoids. Time-dependent increase in size was observed over 6–8 weeks of culture.
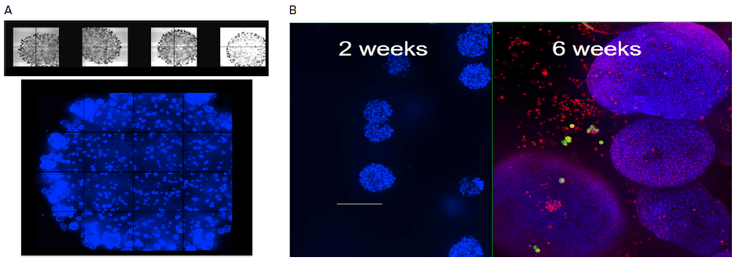
Figure 2. Organoids in Matrigel dome four weeks in culture, TL images (4X) or stained with Hoechst dye (10X). Mean diameters of organoids increased over time. B. Comparison of organoid size, two weeks stained with Hoechst (blue), six week’s samples stained with Hoechst and MitoTracker Orange (red). Organoids were imaged using the confocal option, Z-stack of 23 images 10 μm apart. Maximum projection images shown.
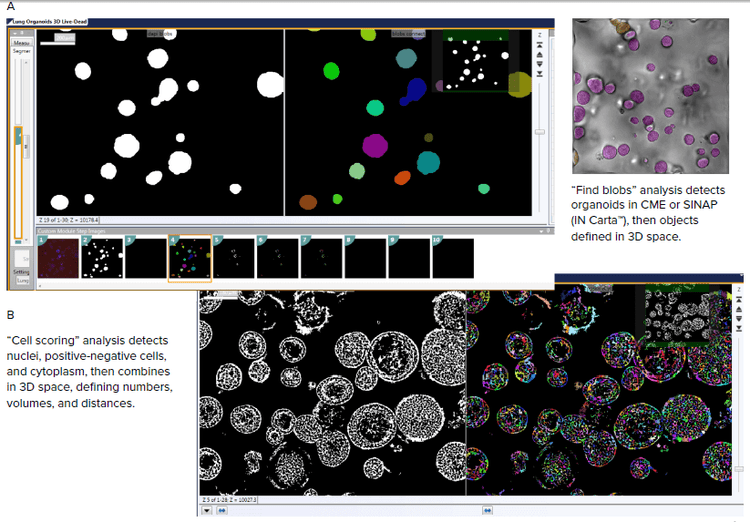
Figure 3. Image analysis recapture complexity of organoid structures using 3D image analysis. Organoids (A), cells or sub-cellular structures (B) can be detected, counted, and characterized using the Custom Module Editor. Analysis performed in each Z-plane, and then software combined the information to give measurements in 3D space.
Measurements of phenotypic changes and compound effects
Organoids provide a very useful tool for disease modeling and assessment of compound effects. Automated imaging and analysis of organoids are important for quantitative assessment of phenotypic changes in organoids, and increases throughput for experiments and tests. 2D analysis of projection images can be used for the highthroughput assays. Example below shows Live-Dead analysis of control organoids and organoids treated with inflammatory cytokines and cytotoxic agents.
Confocal Imaging and 3D image analysis are especially useful for capturing complexity of 3D biological assays. 3D organoids have a hollow appearance, with a lumen or cavities inside, and are more easily penetrated by light, allowing “imaging through” multiple microtissues imbedded into Matrigel. Some anti-cancer drugs, e.g. Ibrutinib, that are used to treat leukemia, have been shown to cause lung toxicity effects. We have tested toxicity of anti-cancer drug Ibrutinib using 3D lung organoids.
Interestingly, significant cytotoxicity was observed at EC50 \~3 μM of Ibrutinib, while a toxicity effect was observed with same cells in 2D culture only at 100 μM concentration. This result may indicate a greater sensitivity of 3D models to cytotoxic effects of selected drugs which highlights the importance of using more complex models for evaluation of compound effects.
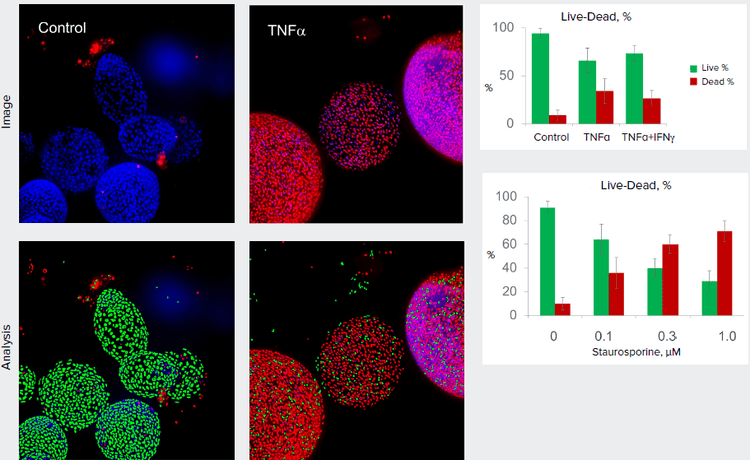
Figure 4. Organoids were treated for three days with 5 nM of TNFα. Organoids stained with Hoechst (blue, nuclear stain) and EtHD (red, stain for dead cells) and imaged with 10X magnification, maximum projections. Images were analyzed in 2D for the counts of live and dead cells.
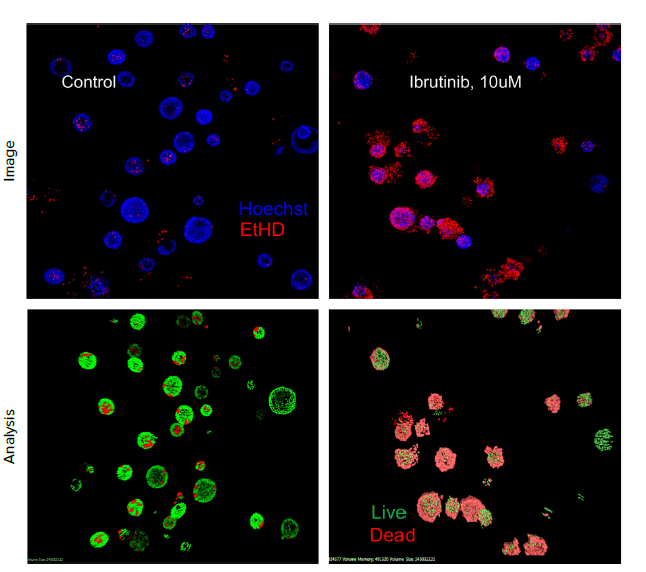
Figure 5. After six weeks of development, organoids were treated with different concentrations of Ibrutinib for 72 h. Organoids were then stained with EtHD-1 to detect dead cells and imaged using ImageXpress Confocal HT.ai system. Numbers of EtHD-1 positive (dead) and negative (live) cells were counted using 3D analysis.
Conclusion
- 3D lung organoids were successfully formed and developed using Stem Cell Technology protocols. Development and growth of organoids were monitored in Matrigel by automated imaging that allowed evaluation of organoids size, volume, and complexity.
- Confocal imaging in combination with 3D analysis allowed quantitative characterization of cellular content as well as count and measurement of cells with different phenotypes within organoids (cell count, live-dead assessment, cell scoring for specific markers, etc.).
- This model can be used for toxicity evaluation of pharmaceutical drugs and other compounds.