
Application Note
Monitor for endotoxin with the SpectraMax ABS Plus Microplate Reader
- Sensitive endotoxin detection to 0.001 EU/mL
- Software protocol with automatic data reduction and standard curve plotting
- Data variability and r values well below required limits
Joyce Itatani | Applications Scientist | Molecular Devices
Cathy Olsen | Sr. Applications Scientist | Molecular Devices
Introduction
Monitoring for contaminants is a critical step during the production process in the pharmaceutical and medical device industries. A frequent contaminant, endotoxin, can cause fever, inflammation, headache, nausea, and even death. Found in the cell wall of gram-negative bacteria, endotoxin has routinely been detected using the sensitive and specific Limulus amebocyte lysate (LAL) assay. In the presence of endotoxins, LAL, obtained from the horseshoe crab Limulus polyphemus, coagulates through an enzymemediated cascade, which can be quantified via gel clot, turbidimetric, or colorimetric tests.
The PYROGENT™-5000 Kinetic Turbidimetric LAL Assay from Lonza is a kinetic assay in which LAL coagulation is monitored as an increase in turbidity over time, which can be measured kinetically on an absorbance plate reader. The more endotoxin is initially present, the shorter the onset time (also known as reaction time), the time required for the absorbance of a sample to increase a specified amount above its initial value. A standard curve of onset time versus standard concentration is run to calculate the amount of endotoxin present in a sample. The assay can be used to detect levels of endotoxin from 0.001 to 10 endotoxin units (EU)/mL.
Materials
- PYROGENT-5000 Kinetic Turbidimetric LAL Assay (Lonza cat. #N383 (100 tests), N384 (200 tests))
- 96-well clear LAL Reagent Grade™ Multi-well Plates (Lonza cat. #25-340)
- Pyrogen-free Dilution Tubes (Lonza cat. #N207)
- LAL Reagent Water (Lonza cat. #W50-100)
- SpectraMax® ABS Plus Microplate Reader (Molecular Devices cat. #ABS PLUS)
Methods
The SpectraMax ABS Plus reader was heated to 37ºC prior to setting up the assay.
Reconstitution of reagents
- Control Standard Endotoxin (CSE) was reconstituted with the volume of LAL reagent water (LRW) indicated on the Certificate of Analysis included with the kit to yield a solution of 100 EU/mL. The vial was vortexed for 15 minutes.
- LAL Reagent was reconstituted with LAL Reconstitution Buffer according to kit instructions.
Standard Curve
- A solution containing 10.0 EU/ml endotoxin was prepared by adding 0.1 ml of the 100.0 EU/mL endotoxin stock into 0.9 ml of LAL Reagent Water.
- A 1:10 dilution series, beginning with the 10 EU/mL standard and ending with a 0.001 EU/mL standard, was made in LRW for the standard curve.
- 100 μL of each endotoxin standard was added to triplicate wells of an LAL Reagent Grade Multi-well Plate.
- The microplate (containing standards) was incubated at 37ºC in the preheated SpectraMax ABS Plus reader for 10 minutes.
- 100 uL of LAL reagent was then added to the wells containing the endotoxin standards, and the kinetic read was started immediately, using the preconfigured Kinetic Turbidimetric LAL protocol in SoftMax® Pro Software, with instrument settings shown in Table 1.
Total run time: 01:40:00
Interval: 00:01:00
Table 1. Instrument settings for kinetic turbidimetric assay on the SpectraMax ABS Plus reader. A kinetic turbidimetric LAL protocol in SoftMax Pro Software contains optimal settings and automates the analysis of results. (MeanValue = Mean onset time)
Data analysis
In the Data Reduction dialog in SoftMax Pro Software, parameters were set at default values except for the following:
- Set first data point to zero
- Kinetic Reduction: Onset Time, with Onset OD 0.03
Average onset times were calculated by the software and plotted vs. endotoxin standard concentration.
Results
Kinetic traces of representative standards ranging from 0.001 to 10 EU/mL are shown in Figure 1, with onset times indicated as vertical red lines. Mean onset time for each standard was plotted vs. standard concentration by SoftMax Pro Software (Figure 2).
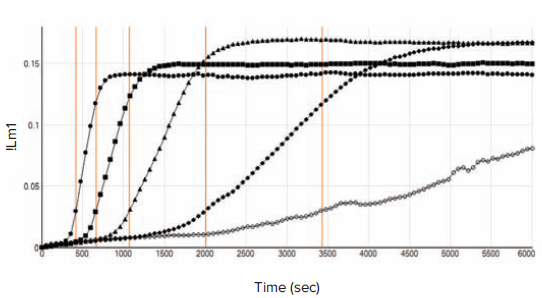
Figure 1. Kinetic traces of representative standards. Vertical red lines indicate onset times (onset OD = 0.03).
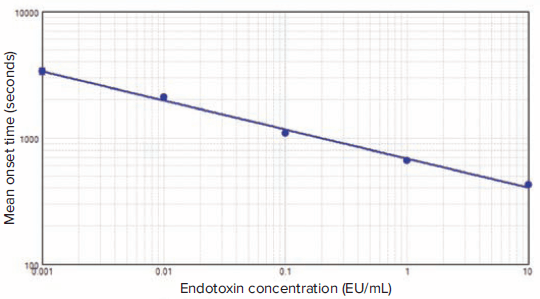
Figure 2. Endotoxin standard curve. Mean onset time (seconds) vs. endotoxin standard concentration was plotted using the log-log curve fit in SoftMax Pro Software (n = 3, r2 = 0.996).
According to the PYROGENT-5000 assay manual, performance requirements include the following:
- The absolute value of the correlation coefficient (r) of the calculated standard curve should be ≥0.980.
- The %C.V. of the reaction times for the replicates should be less than 10%.
For the standard curve generated using the SpectraMax ABS Plus reader, r = 0.998, exceeding the required value of 0.980. The %C.V. calculated by SoftMax Pro Software for standards in triplicate was less than 10% for each standard (Table 2). Equivalent results were obtained using the SpectraMax Plus 384 Microplate Reader (data not shown).
Table 2. Values calculated in the standards group table in SoftMax Pro Software. %C.V. for each standard was less than 10%, meeting the required reproducibility as indicated in the PYROGENT-5000 assay kit manual.
Conclusion
The Limulus Amebocyte Lysate (LAL) test is recognized by the U.S. Pharmacopeia (USP) for determining endotoxin levels in many drug and device products. The quantitative, kinetic PYROGENT-5000 assay offers labs a high-throughput method to test samples for the presence of endotoxin and can detect levels as low as 0.001 EU/mL, making it an effective way to ensure safe endotoxin levels for a variety of sample types. The SpectraMax ABS Plus reader and SoftMax Pro Software, along with the PYROGENT-5000 assay, offer a streamlined workflow for endotoxin testing with automatic calculation of results using a preconfigured protocol in SoftMax Pro Software.