
Cell Counter
At the Cell Counter: Jurkat Cells
Established in the late 1970s from the peripheral blood of a young boy with T-cell leukemia, Jurkat cells are an immortalized T-lymphocyte cell line. They have been used to study T-cell leukemia, T-cell signaling, and cancer cell sensitivity to drug treatments. These small, round cells grow readily in suspension and have also been used as a model system for various cell viability and apoptosis assays as demonstrated here.
View StainFree Cell Detection Webinar
Download StainFree Cell Detection Application Note
Download eBook: Count Cells Like a Pro
Figure 1: StainFree cell counts

Jurkat cells were imaged using the SpectraMax MiniMax 300 Imaging Cytometer, and cells were identified using the predefined ‘CellsC’ setting. Shown on the left are the original transmitted light images and on the right are the same images with purple masks indicating cells identified by the software. Two cell densities are shown: 50,000 and 1562 cells seeded per well of a 96-well plate.
Figure 2: StainFree cell counts vs. fluorescent cell counts
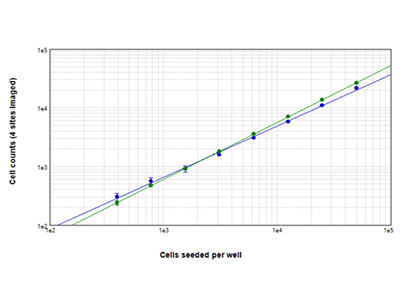
Jurkat cells seeded at densities ranging from 390 to 50,000 cells per well were counted using StainFree technology (blue dots), or they were stained with EarlyTox™ Live Cell Assay dye and green fluorescent cells were counted (green dots). Cell counts obtained with both methods agreed closely across the entire range of cell densities. Cell counts were measured for four sites imaged per well.
Figure 3: EarlyTox™ Live/Dead Assay
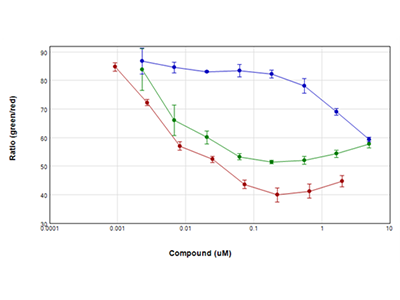
Jurkat cells were treated with serial dilutions of staurosporine (red plot), camptothecin (green plot), and etoposide (blue plot) for 24 hours and then assayed using Molecular Devices EarlyTox Live/Dead Assay Kit. Live cells were stained with calcein AM (green dye), and dead cells were stained with ethidium homodimer (red dye) and read on a SpectraMax i3x Multi-Mode Microplate Reader using the WellScan feature, which performs multiple reads spaced regularly across the well. Results were plotted as ratio of green/red vs. compound concentration. All compounds used in this experiment caused a dramatic reduction in cell viability within 24 hours.
Figure 4: EarlyTox Caspase-3/7 NucView™ 488 Assay
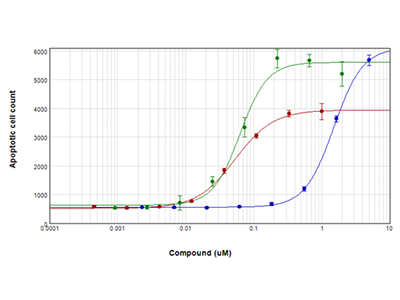
Jurkat cells were treated with staurosporine (red plot), camptothecin (green plot), or etoposide (blue plot) for 28 hours and then assayed for apoptosis using the EarlyTox Caspase-3/7 NucView 488 Assay Kit. Cells expressing caspase-3/7 were stained green fluorescent and were imaged and counted using the SpectraMax MiniMax 300 Imaging Cytometer.
Tip:
Thanks to their rounded morphology and uniform size, Jurkat cells are fairly easy to image and count. For StainFree cell counting, the predefined ‘CellsC’ in SoftMax Pro Software works great.
Jurkat Cells Analysis Toolkit
- SpectraMax ® i3x Multi-Mode Microplate Detection Platform
- SpectraMax ® MiniMax™ 300 Imaging Cytometer
- SoftMax ® Pro Software
Analysis type: Discrete Object Analysis
Wavelength for finding objects: TL
About StainFree Cell Detection Technology
Imaging cell-based assays typically requires the use of fluorescent probes that can be toxic to living cells or may only function in fixed cells. A label-free method for analyzing cell counts and cell confluence enables researchers to quantitatively monitor cell proliferation and health without time-consuming workflows that may disrupt cell viability.
The SpectraMax i3 Multi-Mode Microplate Platform with MiniMax 300 Imaging Cytometer uses unique, patent-pending StainFree Cell Detection Technology that allows you to perform cell proliferation, cytotoxicity, and other assays without nuclear stains like DAPI, which intercalates with DNA, or live cell dyes that are actually toxic to cells in the long term.